New cancer cases have doubled since 1990, with more than 18.5 million diagnoses in 2023. Even as innovation in cancer treatments has advanced, it is now estimated that around one in five people will develop a form of cancer during their lifetime. In the quest to outsmart the various forms of the disease and improve quality of life among cancer patients, teams at GSK are researching multiple technologies, including antibody-drug conjugates (ADCs), which have become a promising option for more targeted treatment. In this article, leading experts explain the science behind this growing field of oncology.
Cancer is unrelenting. Tens of millions of people develop a form of the disease each year and despite a new era of breakthrough therapies, lifestyle-driven risks including diet and alcohol consumption, alongside a global ageing population, continue to drive an upward trend in cancer rates. In 2023 alone, cancer claimed more than 10 million lives and the number of cancer diagnoses is forecast to increase 60% from current levels by 2050.
But equally unrelenting are scientists in their efforts to treat the disease more effectively.
For over a century, radiotherapy and chemotherapy have remained the foundational treatments in oncology by destroying cancerous cells using radiation and anti-cancer drugs, respectively. But they do not discriminate between cells and can harm healthy tissues as a result, as well as cause undesirable adverse side-effects such as organ injury, nausea and hair loss. Cancers may also develop resistance to them.
To overcome these challenges, a range of new therapies are being developed with greater precision and potency in mind, such as small-molecule inhibitors that penetrate cancer cells and alter their activity, and immunotherapies, which boost a person’s immune system to more effectively target cancer cells.
But the most rapidly growing technology within oncology trials is antibody-drug conjugates (ADCs) — therapies designed to track down cancer cells and deliver chemotherapy agents, known as a payload, directly into them to destroy the cells.
“ADCs are engineered to target proteins that are highly expressed on the surface of cancer cells, which allows them to deliver the payload more precisely and limit impact on healthy cells,” said Ken Hance, Vice President, Oncology Extracellular Targeted Cancer Therapeutics Research at GSK, who helps oversee the development of next-generation cancer treatments. “Our aim is to enhance the effectiveness of treatments and the durability of response, while working to minimise adverse side-effects.”

ADCs: Innovatively destroying cancer cells
The development of ADCs has expanded rapidly over the past decade, driven by research into their safety profile and composition as well as by technology such as artificial intelligence (AI). These findings are further shaping the discovery and design of new ADCs such that they can be used to treat more cancer patients.
Today, more than 17 ADCs have been approved globally, and with more than 200 distinct types of cancer known to date, several more are being studied to treat a wider range of cancers with precision, including solid tumours.
Cancers are made up of many different cancer cells with different genetic characteristics. This makes cancer hard to treat because a treatment that kills some tumours may not work on others.
"Even within a single type of cancer, tumours can differ significantly from patient to patient. This genetic diversity of tumours affects how they grow, spread and respond to treatment and this continues to shape oncological research,” said Hance. “Right now, we're at a stage where we're able to identify some patients for whom ADCs are a fit. The goal is to expand that reach so more people can be matched to the therapies most likely to help them.”
How do ADCs work?
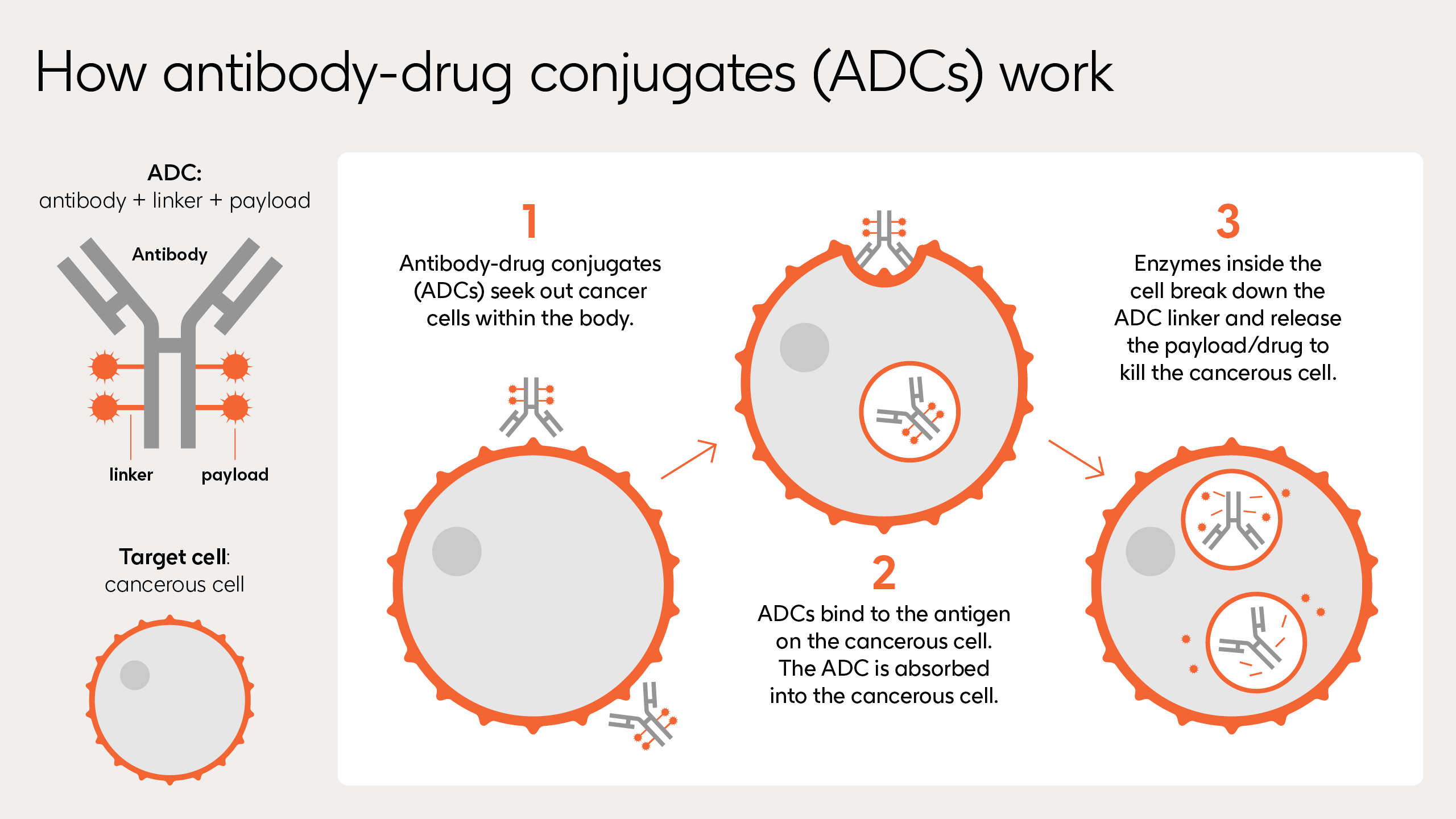
When ADCs are injected into the body, they seek out their target tumour by identifying a unique protein that sits more readily on the surface of the cancerous cells than normal cells, known as an antigen.
Once they find their target cancer cells, ADCs attach, enter and act using three key parts:
- Monoclonal Antibody: Identifies and locks itself onto the tumour cells which possess the target antigen.
- Linker: Connects the monoclonal antibody and payload, ensuring the chemotherapy drug is released at the right time.
- Payload: Carries the chemotherapy agents to destroy the cancerous cell once inside.
When the monoclonal antibody binds onto the cancerous cell, the ADC is internalised by the cell and once inside, enzymes break down the linker to release the payload – a chemotherapy drug. The drug disrupts the cancer cells’ ability to carry out critical functions, with the aim of killing the cell.
As with all medicines, ADCs can cause side effects, although these vary from person to person. In clinical trials, some people have experienced adverse effects such as changes to vision, diarrhoea, nausea, lung inflammation, and haematological toxicity, where someone may have symptoms such as fatigue or shortness of breath due to low levels of blood cells. New studies are exploring ways to minimise this potential impact.
The search for longevity
ADCs provide a different approach to targeting cancer and are being trialled and adopted into clinical practice across different stages of treatment.
“ADCs are being used all the way from neoadjuvant — before patients get surgery — and very early-stage disease, all the way to very, very late lines of the disease,” said Eric Richards, Senior Vice President and Head of Medicine Development Leaders for Oncology at GSK, who leads oncology drug development, including for ADCs.
The technology is also being researched to aid a future where patients with few choices of therapies or a high risk of relapsing have more options available to them.
“ADCs have shown benefits in the recurrent setting for gynaecological cancers,” said Dr. Kathleen Moore, the deputy director and director of Phase 1 oncology trials at the Fred & Pamela Buffett Cancer Center at the University of Nebraska Medical Center.
People with gynaecological cancers experience high relapse rates, meaning current treatments do not adequately meet patients’ needs. Dr. Moore has worked with hundreds of patients and says she has seen promise in early-stage clinical trials with ADCs. Researchers are carefully studying potential side effects, including short‑ and long‑term toxicity, to better understand how these treatments can be used effectively across broader patient populations. “In general, patients feel pretty good, but we have to be cautious with ADCs as we start to really implement these into practice,” Dr. Moore told GSK.
When developing the next generation of the technology, scientists are also focusing on refining linkers, testing different payloads and the use of multiple antibodies to adapt therapies to become more precise and powerful. “I think there's a tremendous opportunity here,” said Moore.
Teams at GSK are also trying to understand the long-term effectiveness.
“There are a lot of drugs that can shrink a tumour initially, but the goal is trying to come up with a combination or sequence [of therapies] that makes that response durable, durable, durable. We need it to just keep on lasting,” said Richards.

The big challenges: Dosing, efficacy and efficiency
ADCs have shown promise, but we are still in the early days of grasping their full potential and several questions need answers around dosing, efficacy, and safety.
“ADCs must be understood thoroughly — where they work, how they work with variations of payloads and with different tumour antigens, and patients’ quality of life following the therapy,” explained Richards.
AI is helping teams at GSK tackle some of these challenges by providing insight into what happens when ADCs reach their target proteins. For example, it can help analyse patterns around how cancer cells interact with the chemotherapy drug, such as specific sensitivities or resistance between the two. It can also potentially identify the patients most likely to respond to certain ADCs.
AI is also a powerful addition to the manufacturing of ADCs, as the process can be complex, Richards highlighted. “AI has a very special place within our drug development paradigm,” he said. “You can take bigger bets possibly with a better quantification of your risk. AI and machine learning may be able to help to do that.”
Looking further ahead, the hope is to tailor treatments when patients have multiple target proteins on their tumour tissues — a common occurrence in cancers — and personalise them with the use of genomics to understand a tumour more specifically, to choose the treatment that will be most effective.
And a big concern, as with any treatment, is resistance. This is commonly observed among patients when the cancer stops responding to the chemotherapy drug used in the ADC. But patients can experience resistance for a few different reasons, including whether the cancer cells actively push the payload out, or if surface antigen mutates and no longer binds the ADC.
“What we're endeavouring to do is understand what's the next therapy that directly addresses that resistance mechanism, understanding what's causing the cancer to progress,” Richards added. “Trying to come up with combinations or sequences of drugs that lead you to the most durable effect – that's really the end goal.”
ADCs at a glance
First ADC approved for medical use:
2000ADCs currently approved globally:
>17ADC candidates in clinical development:
>200