Global health
We are changing the trajectory of high burden diseases in lower income countries, with a focus on infectious diseases.
Where you live continues to define your chances of enjoying a healthy life.
Amongst the biggest drivers of morbidity and mortality in lower income countries are tuberculosis (TB), malaria, HIV, anti-microbial resistance (AMR), as well as Neglected Tropical Diseases (NTDs). We believe that focusing on prevention and treatment of these high burden infectious diseases is a critical part of the global response to address health challenges in lower income countries.
Our rich heritage as a leader in global health gives us a unique and important role to play in improving health for patients around the world. By using our science, technology, talent and partnerships, we’ll deliver health impact at scale.
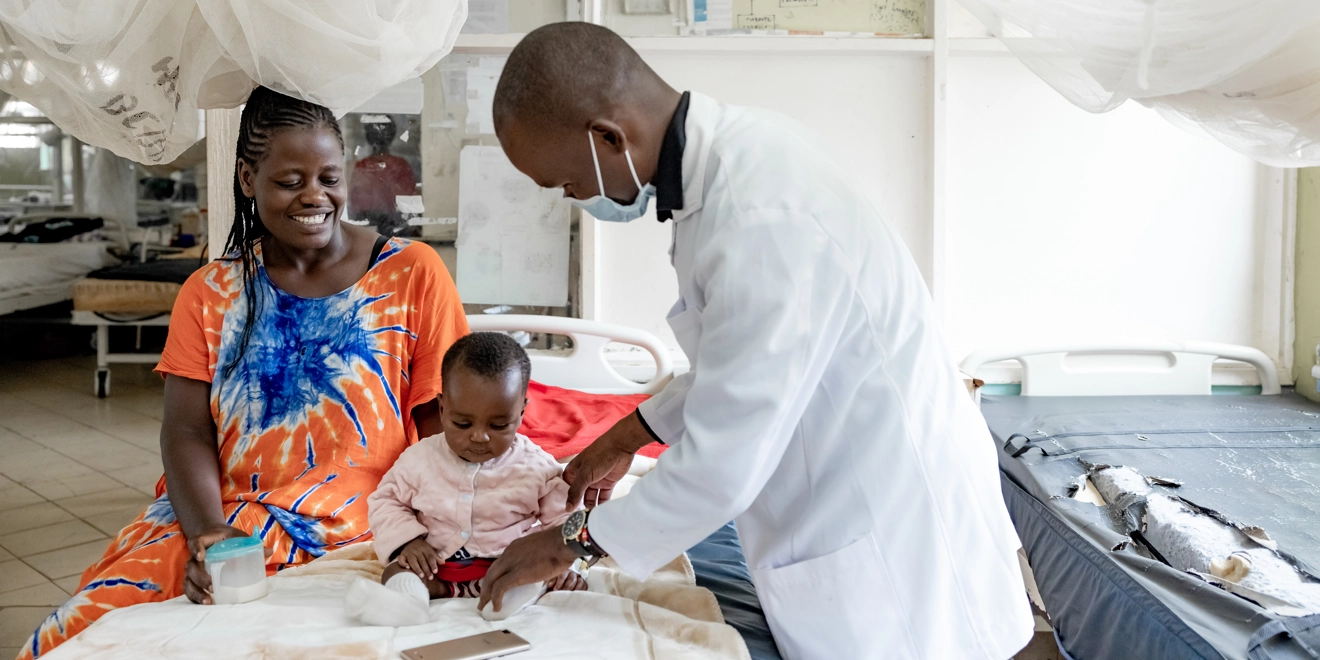
Spotlight on our global health impact
Our focus
We have a dedicated global health team of more than 200 outstanding people and our approach is three-fold to ensure we deliver on positively impacting the health of more than 1.3 billion people in lower income countries.1
Our voices series
Hear from people and communities living in lower income countries about how infectious diseases such as malaria and TB affect them, and their loved ones.
Health care professionals
Dedicated healthcare professionals working in lower income countries share their experiences of helping prevent, diagnose and treat people with TB and malaria.
Our impact partnerships
Our impact partnerships strengthen health systems, drive sustainable change and ensure our innovation reaches patients who need it most.
Our R&D
Developing new medicines and vaccines against high burden infectious diseases in lower income countries.

The Climate and Us series
Why stopping typhoid in lower income countries starts with typhoid.
References
- The methodology for calculating our patient reach impact is derived from a detailed product level analysis of actual sales and long-range forecasts by individual countries. It purely accounts for the number of people receiving a GSK product (reach), not the outcomes or benefits they derive. The analysis uses conservative assumptions to ensure people are only counted once, even if they experience two or more of GSK’s vaccines or medicines during the 10-year period. The methodology and the data have been reviewed and validated externally. Budget phasing is not linear across the 10-year period.


